InnovATEBIO National Center for Biotechnology
InnovATEBIO's Biomanufacturing Hub
View InnovATEBIO Hubs Here

Today, bioprocessing is best understood to be about the manufacture of biotherapeutics. That may sound as though bioprocessing deserves a pat on the...
View Here
Regenerative medicine evolved from medical research rather than from drug-development companies, and technologies that worked in clinical settings...
View Article Here
If you want to “clean up” in the recombinant protein business, you might want to start by sweeping away process-related impurities, specifically, host...
View article here

Article examines the manufacturing challenges involved in the development of viral vector based gene therapies
View Genetic Engineering News

Thirty-five years on from the FDA’s approval of a first monoclonal antibody, these biologics account for nearly a fifth of the agency’s new drug...
View Here

In January 2020, the United States Food and Drug Administration (FDA) released several new or updated guidance documents pertaining to human gene...
View Article Here
Manufacturing Challenges
Despite validation of AAV-vectored gene therapy as an effective treatment modality across multiple disease indications,...
View Here

New and improved platforms for producing viral vectors are emphasizing scalability, customizability, purity, and availability
View article here
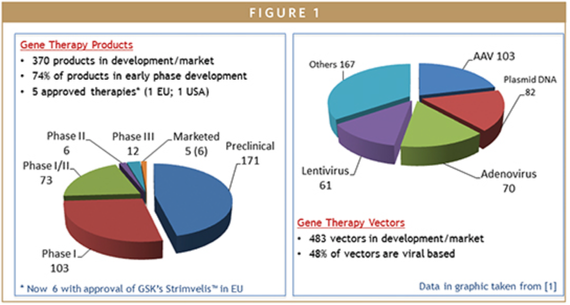
Significant advances in the specific targeting of delivery vectors and the increased therapeutic efficacy of such vectors for gene delivery have been...
View Article Here
Antibody–drug conjugates (ADCs) are an emerging class of targeted therapeutics with the potential to improve the therapeutic index seen with...
View Article Here

The CHO cell is at its height of technological prominence thanks to its adaptability to various culture conditions and plasticity in the context of...
View CHO Consortium SBE Special Section


